Background
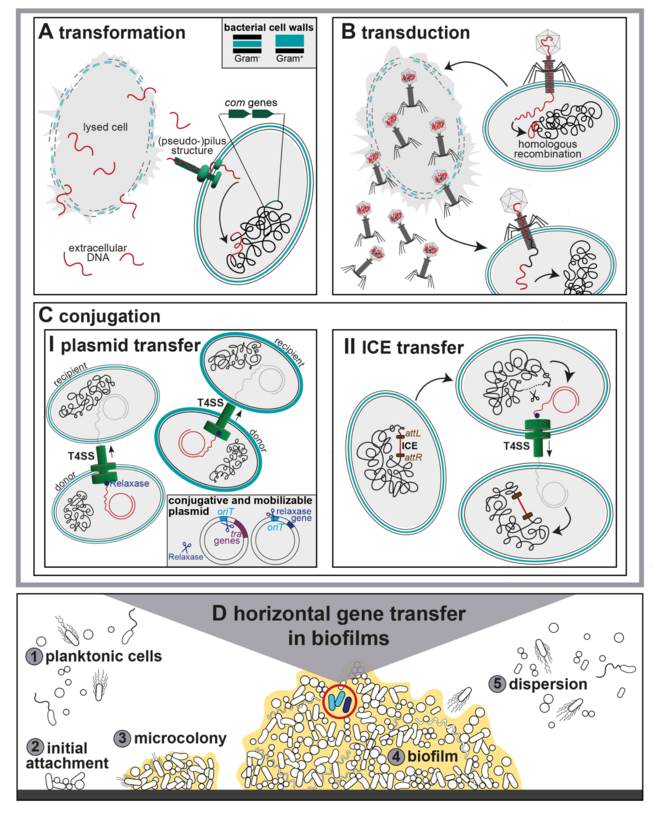
Antibiotic resistance is one of the most pressing challenges in modern medicine. Bacteria can become resistant to antibiotics not only through spontaneous mutations, but also by sharing resistance genes with each other. This exchange can take place even within complex microbial communities known as biofilms.
Biofilms are structured, surface-associated communities of bacteria that form on medical devices such as catheters and implants, as well as on chronic wounds. The protective extracellular matrix surrounding biofilms shields bacteria from antibiotics and environmental stress, making infections extremely difficult to treat. Within these communities, bacteria are in close physical proximity—ideal conditions for genetic exchange.
Bacteria do not only pass genetic information on to their offspring. They can also exchange genes directly between neighboring cells through a process called horizontal gene transfer. The most important mechanism of this process is conjugation. During conjugation, small circular DNA molecules known as plasmids are transferred from a donor bacterium to a recipient cell. These plasmids can carry additional genetic traits, such as antibiotic resistance genes. In this sense, plasmids act as true “rings of power” in the bacterial world, enabling bacteria to rapidly gain new abilities.
The transfer of plasmids requires direct cell-to-cell contact and is mediated by a specialized molecular machine called the Type IV secretion system (T4SS). This complex protein structure forms a molecular tunnel through which the plasmid DNA is transferred as a copy from one cell to another.
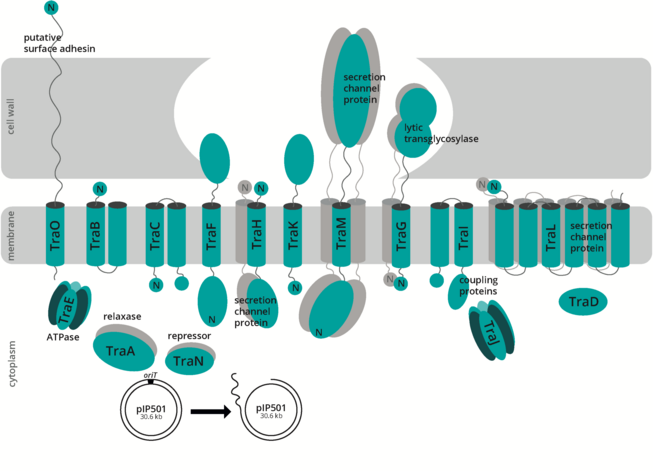
While the mechanisms of conjugation has been extensively studied in Gram-negative bacteria, the molecular mechanisms of plasmid transfer in Gram-positive bacteria are still poorly understood. A key difference between these groups lies in the structure of their cell walls, which strongly influences how genetic material is transferred. Gram-positive pathogens such as Enterococcus and Staphylococcus are among the most common causes of hospital-acquired infections and are increasingly resistant to multiple antibiotics.
Methods and Research Approach
Research in the Grohmann Lab aims to close this knowledge gap by investigating conjugative gene transfer between Gram-positive bacteria in biofilms. A central model system in our studies is the broad-host-range conjugative plasmid pIP501. pIP501 encodes a T4SS and serves as an ideal tool to investigate plasmid-mediated resistance transfer in Gram-positive bacteria.
Using fluorescently labeled derivatives of pIP501, we combine molecular genetics, fluorescence-based reporter systems, advanced fluorescence microscopy, and quantitative microbiological methods to visualize and quantify plasmid transfer events. Conjugative transfer is analyzed in mono- and polymicrobial biofilm models using clinically relevant Gram-positive genera, including Enterococcus and Staphylococcus.
Central questions include how frequently resistance genes are transferred, how this frequency changes under antimicrobial pressure, and how extreme conditions such as microgravity, studied through experiments on the International Space Station (ISS), affect gene transfer dynamics.
Understanding these processes is essential for developing new strategies to limit the spread of antibiotic resistance and to improve the treatment of infections, particularly in clinical environments.